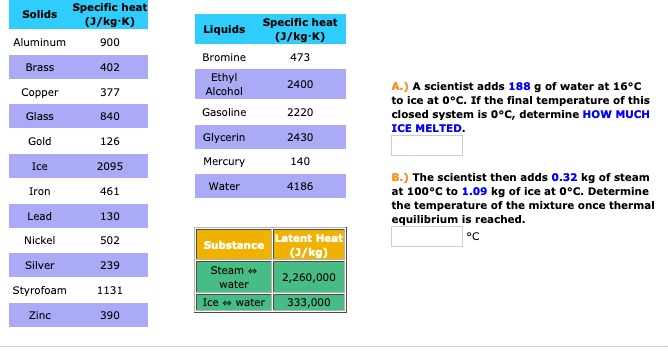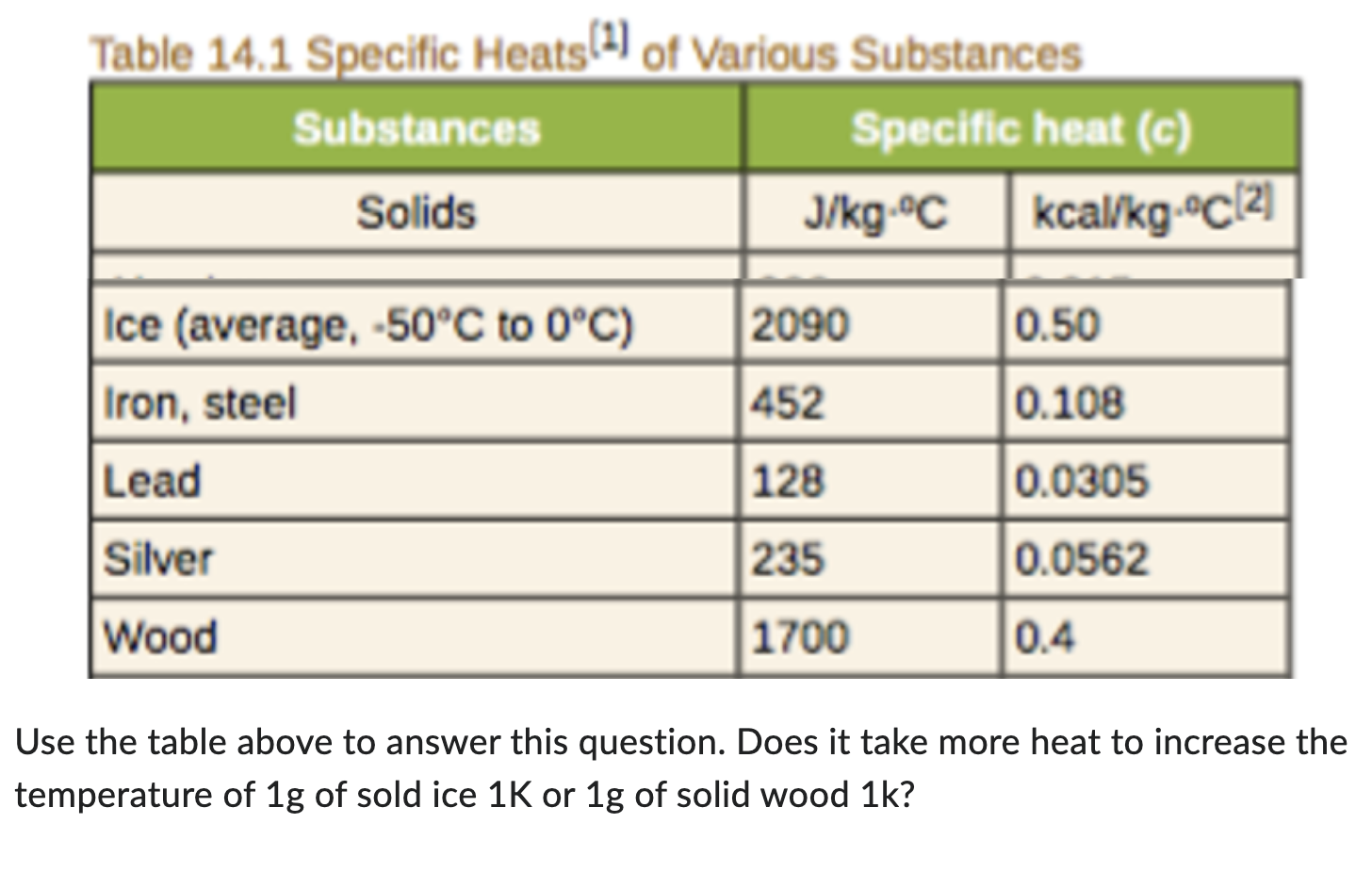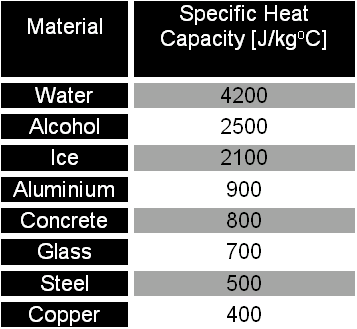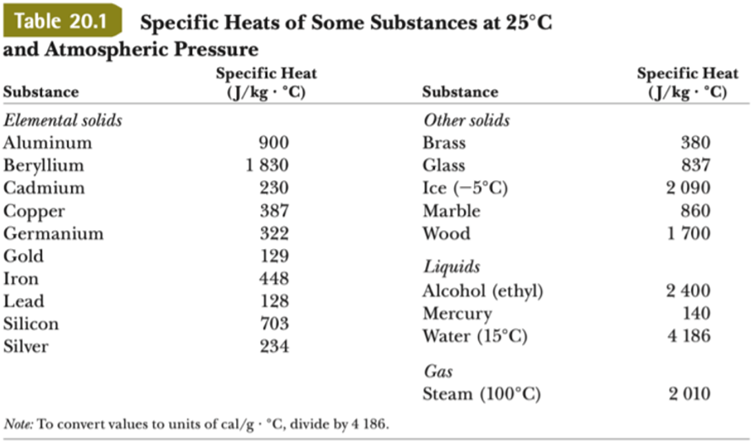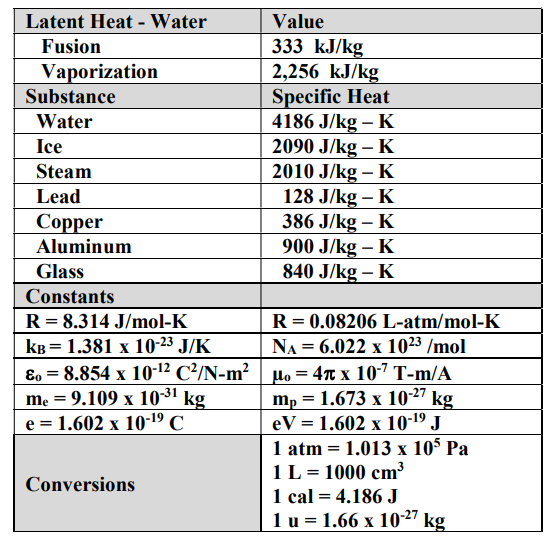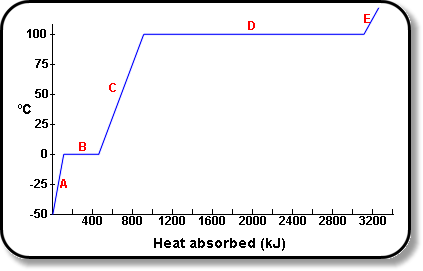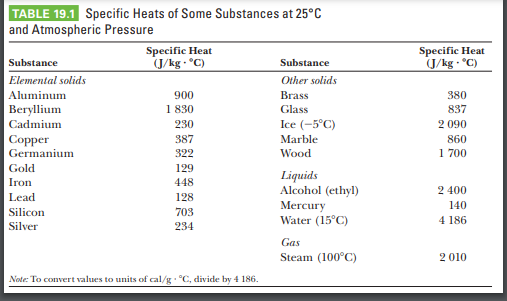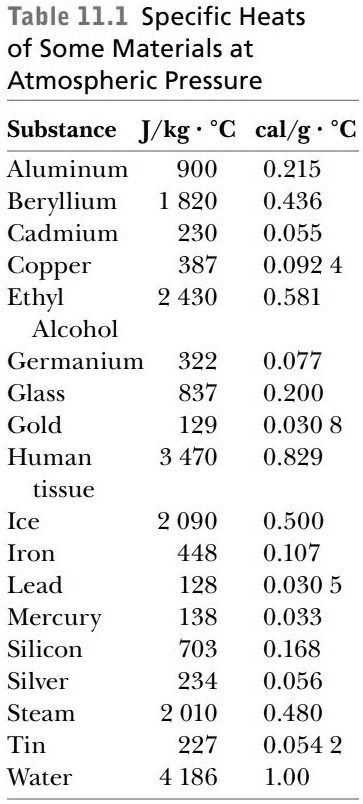
SOLVED: Table 11.1 Specific Heats of Some Materials at Atmospheric Pressure Substance J/kg 'C cal/g Aluminum 900 0.215 Beryllium 820 0.436 Cadmium 230 0.055 Copper 387 0.092 4 Ethyl 2 430 0.581

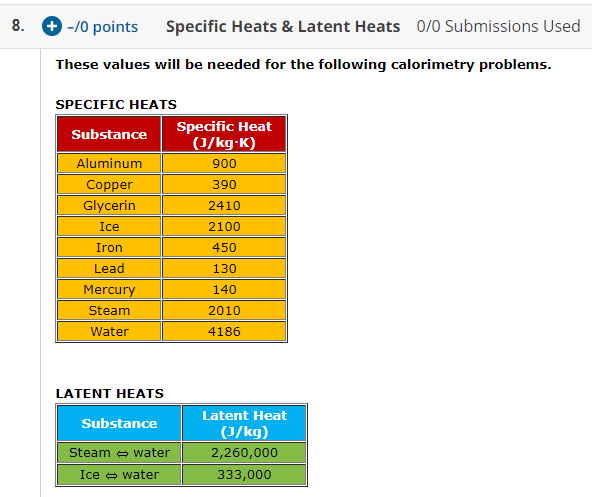
SOLVED: Specific heat capacity of water = 4186 J kg^-1 K^-1 Specific heat capacity of ice = 2090 J kg^-1 K^-1 Latent heat of fusion of ice = 335 kJ kg^-1 Latent
![Specific heat capacity C p [J/(kg·K)] of small samples (E, F, and G) at... | Download Scientific Diagram Specific heat capacity C p [J/(kg·K)] of small samples (E, F, and G) at... | Download Scientific Diagram](https://www.researchgate.net/publication/256115072/figure/tbl1/AS:755605098729495@1557161702871/Specific-heat-capacity-C-p-J-kgK-of-small-samples-E-F-and-G-at-various.png)
Specific heat capacity C p [J/(kg·K)] of small samples (E, F, and G) at... | Download Scientific Diagram

A 0.60 kg sample of water and a sample of ice are placed in two compartments A and B that are separated by a conducting wall, in a thermally insulated container. The

a) Specific heat capacity c (Eq. (11)), thermal conductivity k (Eq.... | Download Scientific Diagram

1 PHYS1001 Physics 1 REGULAR Module 2 Thermal Physics HEAT CAPACITY LATENT HEAT What is cooking all about? ptC_heat.ppt. - ppt download